 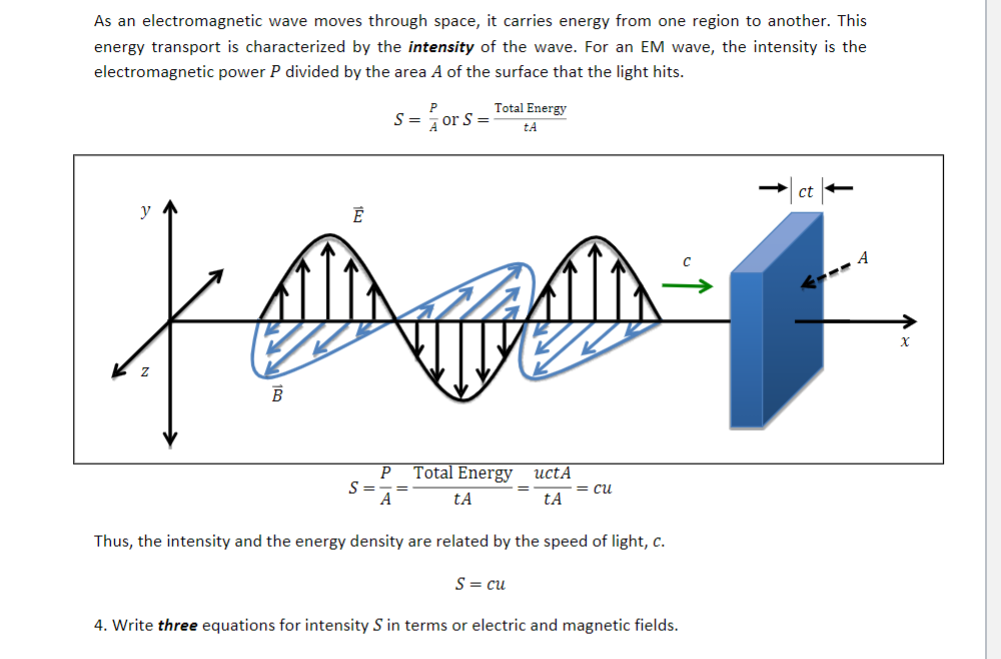
A minimum of 48 photons is needed for the synthesis of a single glucose molecule from CO 2 and water (chemical potential difference 5 × 10 −18 J) with a maximal energy conversion efficiency of 35%. This corresponds to frequencies of 2.42 × 10 25 to 2.42 × 10 29 Hz.ĭuring photosynthesis, specific chlorophyll molecules absorb red-light photons at a wavelength of 700 nm in the photosystem I, corresponding to an energy of each photon of ≈ 2 eV ≈ 3 × 10 −19 J ≈ 75 k B T, where k B T denotes the thermal energy. Very-high-energy gamma rays have photon energies of 100 GeV to over 1 PeV (10 11 to 10 15 electronvolts) or 16 nanojoules to 160 microjoules. 
This minuscule amount of energy is approximately 8 × 10 −13 times the electron's mass (via mass-energy equivalence). Different wavelengths are diffracted at different angles, according to the grating relationship. The Beer-Lambert law relates the attenuation of light to the properties of the material through which the light is traveling. Orders 1 and 2 are shown to each side of the direct beam. The peak intensity of absorbed light falls at about 550 nanometers, right in the center of the green region of visible wavelengths. Green and purple light have more energy, and when you shine them on the potassium you can kick off an electron. When light of a single wavelength, like the 632.8nm red light from a helium-neon laser at left, strikes a diffraction grating it is diffracted to each side in multiple orders. The photon energy of near infrared radiation at 1 μm wavelength is approximately 1.2398 eV.Īn FM radio station transmitting at 100 MHz emits photons with an energy of about 4.1357 × 10 −7 eV. 6.2.2: Potassium requires 2.0eV to eject an electron, and a photon of red light (700nm) only has 1.77eV and so no matter intense the red light is, it can not kick of an electron. E = h c λ eVm where h is Planck's constant, c is the speed of light in m/sec, and e is the electron charge.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
